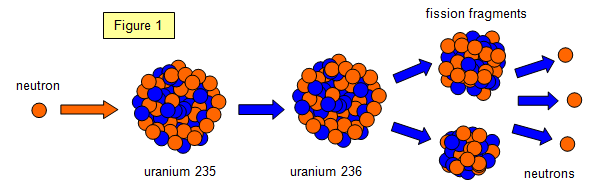
Deuterium is sometimes given the symbol ‘D’ so heavy water can be referred to as D 2O. Unlike a lot of isotopes, H-2 has a special name we can call it besides hydrogen: Deuterium. Heavy water is mostly just like ordinary water (H 2O) but with the regular hydrogen atoms (H-1) replaced by atoms with an extra neutron (H-2). However, different isotopes of the same element can have very different nuclear properties. Because different isotopes of the same element have the same number of protons and electrons, they behave similarly in their chemistry (they may behave slightly differently physically due to their different masses). A small amount of hydrogen (about 0.01%) has one proton one neutron, so it is referred to as H-2. For example, the most common isotope of natural hydrogen has just one proton a no neutron, so it is referred to as H-1. They are named using the letter abbreviation of the element and the total number of protons and neutrons in the nucleus. Isotopes are different versions of elements. While the reactor is running, the vast majority of neutrons are produced by the fission of U-235 in our fuel. This chain reaction can continue if there are enough fissile nuclei in a small enough space, and the neutrons don’t get absorbed by other materials or leak out from that space. This process is the fission chain reaction. Those can hit other nearby uranium-235 atoms and cause those to fission, emitting more neutrons. 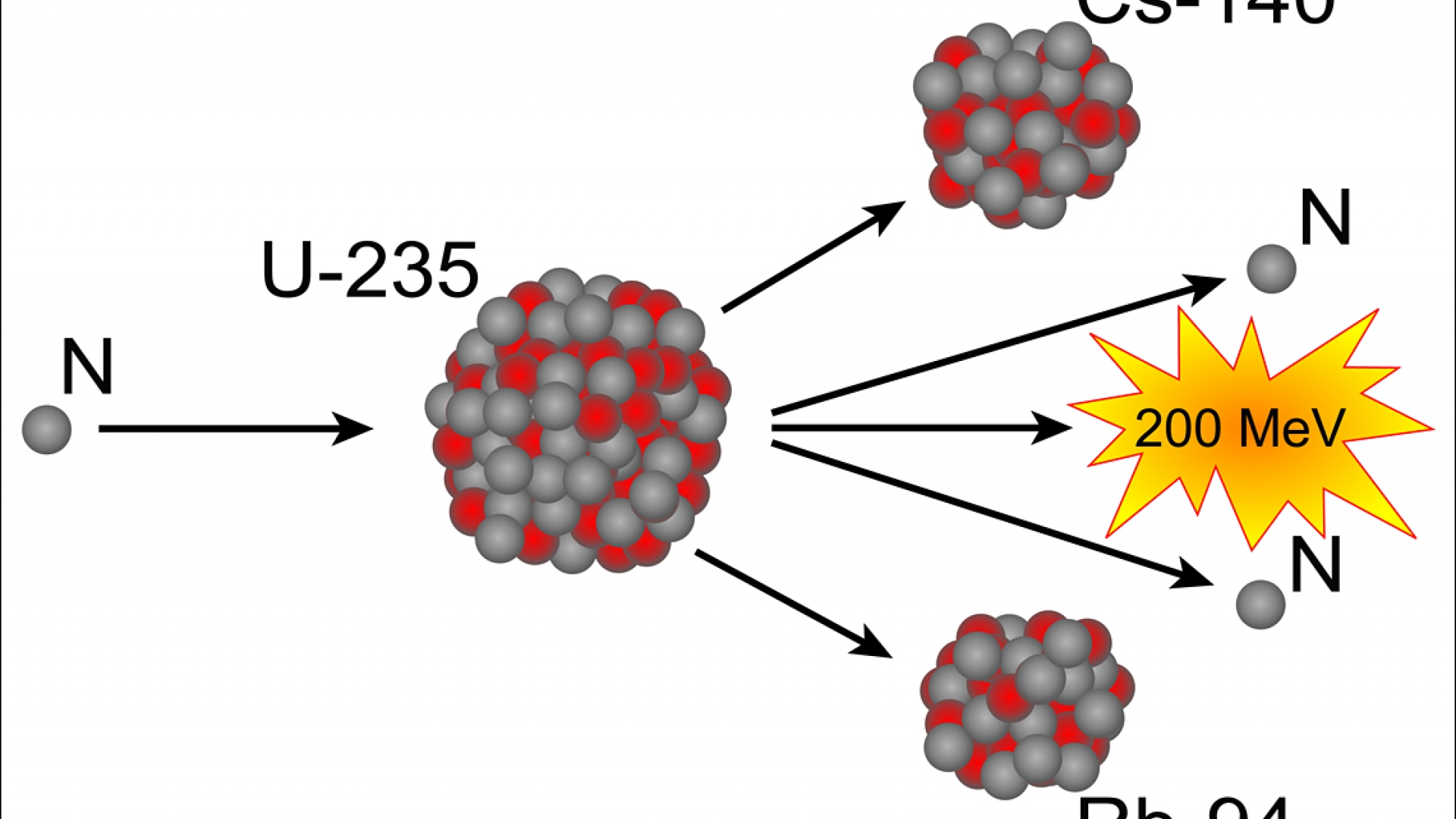
What is a chain reaction?įor example, when a nucleus such as uranium-235 fissions, it emits neutrons. Some isotopes such as californium-252 can spontaneously fission, though most isotopes that are can undergo fission need some stimulation or disruption, such as the absorption of a neutron, in order to cause fission. At the MIT Reactor Lab uranium-235 fissions in the core to produce heat (which we don’t use) and neutrons (which we use for research and experiments). Fission is the nuclear process that involves the splitting of a nucleus.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
